These result from the precipitation in the boiler of feedwater hardness constituents due to heat and interaction of treatment chemicals and from corrosion products in the feedwater.
Boiler water hardness is increased by.
The chelated cations do not deposit in the boiler.
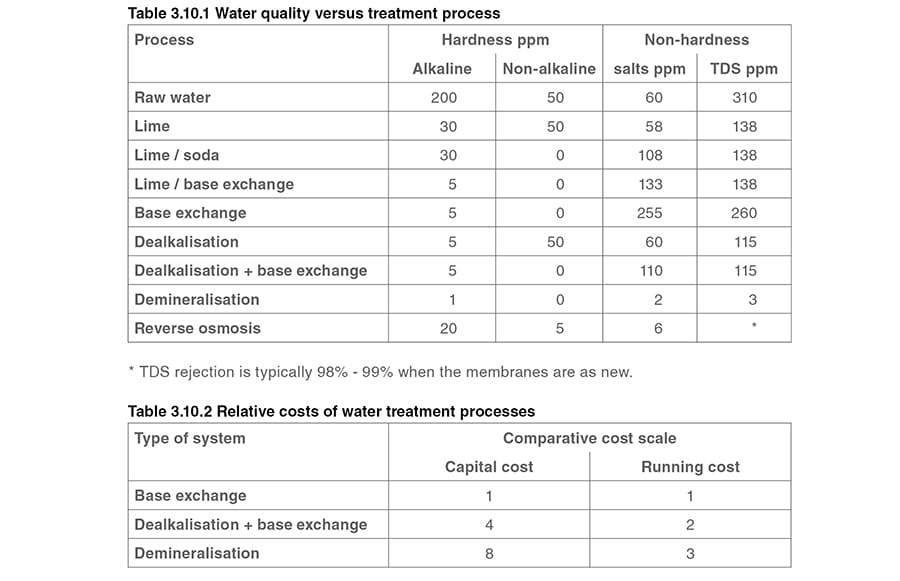
How many times the mineral content which stays in the boiler when steam is produced of the raw water has been concentrated or built up in the boiler.
They get caught inside intricate devices like control valves and pressure regulators.
Contamination of the surfaces of control valves this will affect their operation and reduce their capacity.
This causes a lot of damage and increased maintenance.
Chelants have the ability to complex many cations hardness and heavy metals under boiler water conditions.
Carry over is boiler water that leaves the boiler in the steam but is still water.
Therefore chlorides are used as a measure of boiler water concentrations i e.
Then boiler setpoints were reduced to increase the rate of solids removal and encourage increased soft water makeup into the boiler system until all traces of hardness were no longer entering the boiler feed water system.
They accomplish this by locking metals into a soluble organic ring structure.
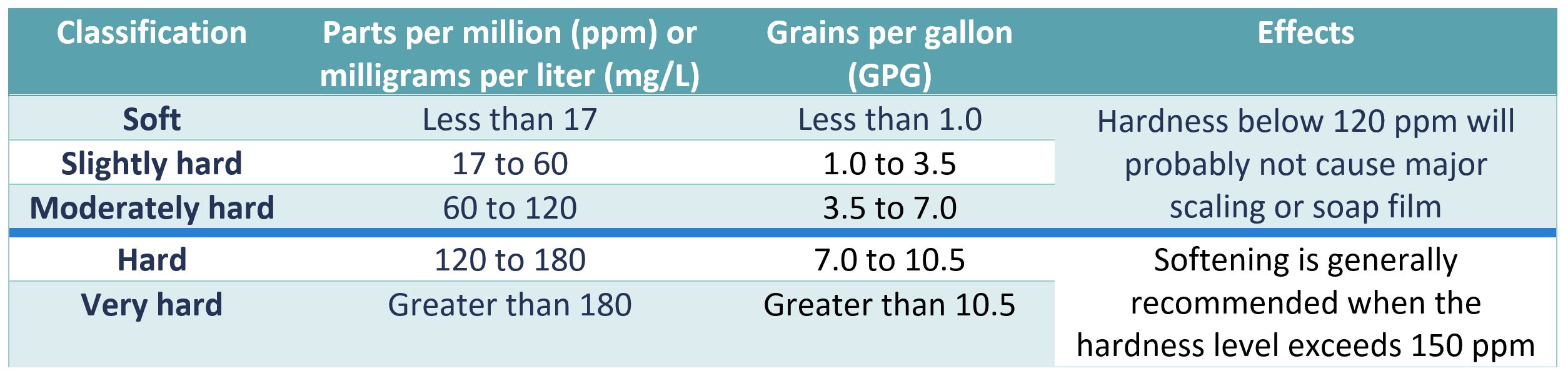
Untreated water even water coming from a municipal water utility can contain dissolved salts which form scale on the heat transfer surfaces as the water is heated.
If the impurities in the boiler feedwater are not dealt with properly carryover of boiler water into the steam system can occur.
Hard water going into a steam boiler can lead to increased scaling and deposit build ups.
This may lead to problems elsewhere in the steam system such as.
Silica ppm sio 2 lt.
Leading to increased carryover.
These impurities leave deposits around the steam system.
The main disadvantage of this deposited.
It carries impurities such as dissolved solids with it.
Total alkalinity ppm as caco 3.